The other lead investigators on the paper, published as a "Breakthrough Manuscript" in Nucleic Acids Research, are Masad Dahma, Ph.D., professor and chair of the Department of Chemistry at McGill University in Montreal, and Scott Stevens, Ph.D., associate professor of molecular genetics and microbiology at The University of Texas at Austin.
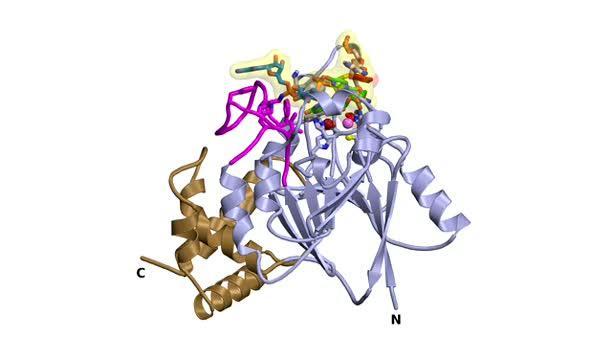
In this movie, the Dbr1 enzyme rotates 360 degrees. Partially inhibiting Dbr1 could represent a new way to treat most cases of amyotrophic lateral sclerosis (ALS), according to a new report.
(Photo Credit: P. John Hart, Ph.D., X-ray Crystallography Core Laboratory, The University of Texas Health Science Center at San Antonio)
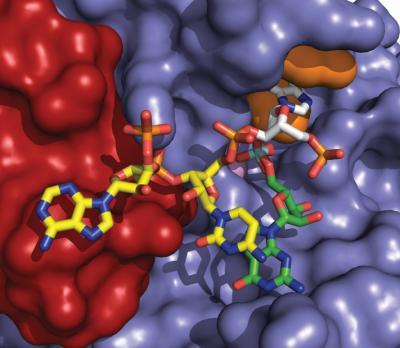
The Dbr1 enzyme (red, blue and orange surfaces) binds to part of an intron lariat (seen as yellow, white and green sticks). This is the first visualization of a Dbr1-intron lariat interaction. Partially inhibiting Dbr1 could represent a new way to treat most cases of amyotrophic lateral sclerosis (ALS).
(Photo Credit: P. John Hart, Ph.D., X-ray Crystallography Core Laboratory, The University of Texas Health Science Center at San Antonio)
Source: University of Texas Health Science Center at San Antonio