Functional loss of limb control in individuals with spinal cord injury or stroke can be caused by interruption of the neural pathways between brain and spinal cord, although the neural circuits located above and below the lesion remain functional.
An artificial neural connection that bridges the lost pathway and connects brain to spinal circuits has potential to ameliorate the functional loss.
Researchers investigated the effects of introducing a novel artificial neural connection which bridged a spinal cord lesion in a paretic monkey that had a unilateral spinal cord lesion at the C2 level.
The first application bridged the impaired spinal lesion and allowed the monkey to drive the spinal stimulation through volitionally controlled power of high-gamma activity in either the premotor or motor cortex and the second application created an artificial recurrent connection from a paretic agonist muscle to a spinal site.
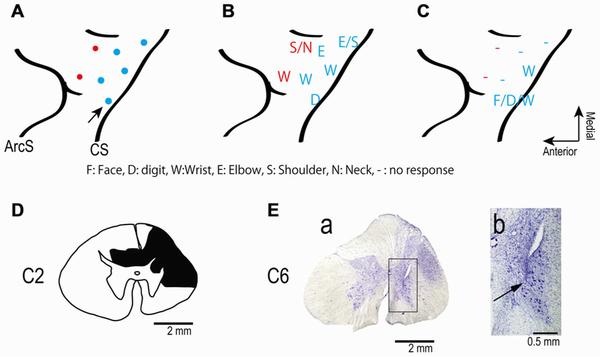
(A) Electrode locations in the motor areas of the lateral aspect of the frontal lobe of the left (contralesional) hemisphere. Electrodes were placed in primary motor cortex (blue dots) and in dorsal premotor cortex (red dots). (B,C) Somatotopic map shows movements evoked from each site in frontal lobe before (B) and after (C) spinal cord injury. The pre-lesion maps were established by ICMS at movement threshold (20–120 μA). The post-lesion maps were established by ICMS at 450 μA on post-lesional day 14. The maps show the region between the central sulcus (CS: diagonal line to the right of each panel) and the arcuate sulcus (ArcS: curved line to the left). Arrow indicates site used in session illustrated in Figure 3. (D) Drawing of the C2 segments showing the extent of the spinal cord lesion (hatched in black). (E) Electrode location in spinal cord. (a) Electrodes were targeted at the ventral horn and intermediate zone of the spinal cord. (b) Higher magnification view of the location of an electrode tip (black arrow). Credit: doi: 10.3389/fncir.2013.00057
This allowed the monkey to electrically stimulate the spinal cord through volitionally controlled brain activity and thereby to restore volitional control of the paretic hand. This study demonstrates that artificial neural connections can compensate for interrupted descending pathways and promote volitional control of upper limb movement after damage of neural pathways such as spinal cord injury or stroke.
Citation: Nishimura Y, Perlmutter SI, and Fetz EE (2013) Restoration of upper limb movement via artificial corticospinal and musculospinal connections in a monkey with spinal cord injury. Front. Neural Circuits 7:57. doi: 10.3389/fncir.2013.00057