The discovery could have implications for understanding diseases such as cancer as well, because cancerous cells tend to move in distinct ways from normal cells.
"It might give us leverage to find out what is unique about cancer cells so we can target them therapeutically and not affect normal cells," Petrie said.
Importantly, these findings could have broader relevance to other biological systems where living cells are enmeshed within and surrounded by an extracellular matrix, such as in biofilms, which are associated with many human infectious diseases.
"This work illustrates how the physical structure of the matrix can influence cellular properties to govern biological function," Koo said. "We are now applying these fascinating principles to further understand how biofilm matrix modulates bacterial virulence to cause oral diseases, such as dental caries."

Penn and NIH researchers have demonstrated a never-before characterized type of cell movement. In this video, a cell's vimentin cytoskeleton (green) pulls the nucleus (red) forward to generate a high-pressure protrusion, called a lobopodia, inside a cell migrating in a physiological 3D extracellular matrix. This time-lapse movie is 30 minutes of time compressed into 4 seconds (450x speed).
(Photo Credit: NIH)
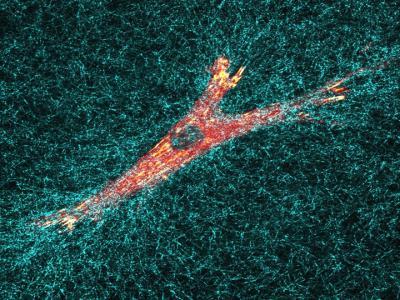
Penn and NIH researchers measured the internal pressure of individual fibroblast cells (in orange) moving through a three-dimensional matrix (in blue). They found that, in this environment, the cells' nuclei operate like an engine's piston to push the cell forward.
(Photo Credit: University of Pennsylvania/NIDCR)
Source: University of Pennsylvania