Biological processes are generally based on events at the molecular and cellular level. To understand what happens in the course of infections, diseases or normal bodily functions, scientists would need to examine individual cells and their activity directly in the tissue. The development of new microscopes and fluorescent dyes in recent years has brought this scientific dream tantalisingly close. Scientists from the Max Planck Institute of Neurobiology in Martinsried have now presented not one, but two studies introducing new indicator molecules which can visualise the activation of T cells. Their findings provide new insight into the role of these cells in the autoimmune disease multiple sclerosis (MS). The new indicators are set to be an important tool in the study of other immune reactions as well.
Inflammation is the body's defence response to a potentially harmful stimulus. The purpose of an inflammation is to fight and remove the stimulus – whether it be disease-causing pathogens or tissue. As an inflammation progresses, significant steps that occur thus include the recruitment of immune cells, the interactions of these cells in the affected tissue and the resulting activation pattern of the immune cells. The more scientists understand about these steps, the better they can develop more effective drugs and treatments to support them. This is particularly true for diseases like multiple sclerosis. In this autoimmune disorder cells from the body's immune system penetrate into the central nervous system where they cause massive damage in the course of an inflammation.
In order to truly understand the cellular processes involved in MS, scientists ideally need to study them in real time at the exact location where they take place – directly in the affected tissue. In recent years, new microscopic techniques and fluorescent dyes have been developed to make this possible for the first time. These coloured indicators make individual cells, their components or certain cell processes visible under the microscope. For example, scientists from the Max Planck Institute of Neurobiology have developed a genetic calcium indicator, TN-XXL, which the cells themselves form, and which highlights the activity of individual nerve cells reliably and for an unlimited time. However, the gene for the indicator was not expressed by immune cells. That is why it was previously impossible to track where in the body and when a contact between immune cells and other cells led to the immune cell's activation. Now the Martinsried-based neuroimmunologists report two major advances in this field simultaneously. One is their development of a new indicator which visualises the activation of T cells.
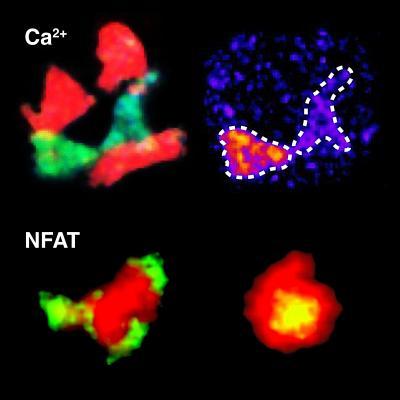
Using a calcium sensor shows that the calcium concentration in T cells (green) changes when it interacts with dendritic cells (red) [top]. The activation of the T cell (red) can be illustrated by the migration of the NFAT signal protein (green) from the cell plasma to the cell nucleus [bottom].
(Photo Credit: MPI of Neurobiology)
These cells, which are important components of the immune system, detect and fight pathogens or substances classified as foreign (antigens). Multiple sclerosis, for example, is one of the diseases in which T cells play an important role: here, however, they detect and attack the body's brain tissue. If a T cell detects "its own" antigen, the NFAT signal protein migrates from the cell plasma to the nucleus of the T cell. "This movement of the NFAT shows us that the cell has been activated, in other words it has been 'armed'," explains Marija Pesic, lead author of the study published in the Journal of Clinical Investigation. "We took advantage of this to bind the fluorescent dye called GFP to the NFAT, thereby visualising the activation of these cells." The scientists are thus now able to conclusively show in the organism whether an antigen leads to the activation of a T cell. The new indicator is an important new tool for researching autoimmune diseases and also for studying immune cells during their development, during infections or in the course of tumour reactions.
In parallel to these studies, the neuroimmunologists in Martinsried developed a slightly different, complementary method. They modified the calcium indicator TN-XXL to enable, for the first time, T cell activation patterns to be observed live under the microscope, even while the cells are wandering about the body. When a T cell detects an antigen, a rapid rise in the calcium concentration within the cell ensues. The TN-XXL makes this alteration in the calcium level apparent by changing colour, giving the scientists a direct view of when and where the T cells are being activated.
"This method has enabled us to demonstrate that these cells really can be activated in the brain," says a pleased Marsilius Mues, lead author of the study which has just been published in Nature Medicine. Until now, scientists had only suspected this to be the case. In the animal model of multiple sclerosis, scientists are now able to track not only the migration of the T cells, but also their activation pattern in the course of the disease. Initial investigations have already shown, besides the expected activation by antigen detection, that numerous fluctuations in calcium levels also take place which bear no relation to an antigen.
"These fluctuations can tell us something about how potent the T cell is, how strong the antigen is, or it may have something to do with the environment," speculates Marsilius Mues. These observations could indicate new research approaches for drugs – or they could even show whether a drug actually has an effect on T cell activation.
Source: Max-Planck-Gesellschaft