Unlike most of our sensory systems that detect only one type of stimuli, our sense of smell works double duty, detecting both chemical and mechanical stimuli to improve how we smell, according to University of Pennsylvania School of Medicine researchers in the March issue of Nature Neuroscience.
This finding, plus the fact that both types of stimuli produce reaction in olfactory nerve cells, which control how our brain perceives what we smell, explains why we sniff to smell something, and why our sense of smell is synchronized with inhaling.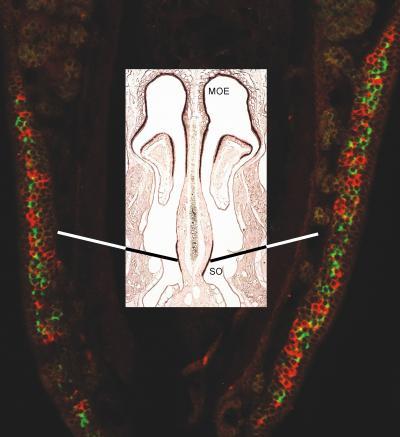 Cross section of olfactory sensory neurons in a mouse nose visualized by fluorescent staining of odorant receptor genes (red and green represent different odorant receptors). MOE: main olfactory epithelium; SO: septal organ. Credit: Huikai Tian, PhD, Minghong Ma, PhD, University of Pennsylvania School of Medicine
Cross section of olfactory sensory neurons in a mouse nose visualized by fluorescent staining of odorant receptor genes (red and green represent different odorant receptors). MOE: main olfactory epithelium; SO: septal organ. Credit: Huikai Tian, PhD, Minghong Ma, PhD, University of Pennsylvania School of Medicine
"The driving force for such synchronization remained a mystery for more than 50 years," says senior author Minghong Ma, PhD, Assistant Professor of Neuroscience. "These results help us understand how the mammalian olfactory system encodes and decodes odor information in the environment."
Researchers tested two different types of stimulation on olfactory neurons in mice: chemical stimuli, such as those used in making perfumes that have almond-like and banana-like scents, and mechanical stimuli, that is pressure carried by air flow to the nostrils while breathing.
The group did this first by puffing a chemical stimulus into the nose. As expected, this produced a reaction in the olfactory neurons, the primary sensory neurons in the nose that perceive odors. Researchers then puffed a solution without the chemical stimuli into the mouse's nose. This also produced a similar, but smaller reaction in the olfactory neurons. By decreasing pressure of the non-odor solution, they also found that the reaction in the olfactory neurons was less, confirming that it was sensitive to mechanical stimulation.
When olfactory neurons respond to odor molecules, they transmit chemical energy into electrical signals, which then trigger a secondary molecular messenger cascade that generates electrical impulses to the brain, signaling that it is smelling something. The group discovered that chemical and mechanical stimuli both resulted in the same messenger molecule, cAMP, which acts like a gatekeeper of reactions in the olfactory neurons.
Although this study was conducted on a mouse model, the researchers tested two different parts of the nose, one that humans have and one that humans do not. The first, the septal organ, is a patch of smell-sensitive tissue on the septal wall of the nasal cavity. The second, the main olfactory epithelium, is a smell-sensitive tissue inside the nasal cavity.
Synchronized Breathing
The septal organ is only about 1 percent the size of the main olfactory epithelium and isn't shared by all mammals. Mice, for example, have a septal organ. Humans do not. But in this study, Ma's group found that 50 percent of the cells in the main olfactory epithelium are sensitive to physical stimuli, suggesting that mechanosensitivity of the olfactory sensory neurons exists in all mammals, even those like humans, without the septal organ.
The mechanosensitivity of our olfactory neurons has two possible functions, suggest the investigators. The first is that it increases our ability to smell, enhancing the detection of odorous molecules in the air. The second is a peripheral drive in the brain to synchronize rhythmic activity, which is the concurrent firing of neurons in the olfactory bulb with breathing.
"The mechanosensitivity may increase the sensitivity of our nose, especially when stimulated by weak odors," says Ma. "It helps the brain make better sense out of odor responses when it integrates airflow information. We still don't know how it happens, but sniffing is essential for odor perception."
Co-authors are Xavier Grosmaitre and Lindsey C. Santarelli, both from Penn, and Jie Tan and Minmin Luo of the National Institute of Biological Sciences (Beijing, China). The National Institute on Deafness and Other Communication Disorders, the Whitehall Foundation, and the University of Pennsylvania Institute on Aging provided funding for this research.
Written from a news release by University of Pennsylvania School of Medicine.